
by Elizabeth Kissling | Dec 29, 2009 | Girls, Internet, Menarche, New Research
Janice Horowitiz’ “Dueling Docs” feature at Huffington Post today is about the issue of girls reaching puberty at increasingly earlier ages than previous generations. Both Dr. Alisan Goldfarb and Dr. Stephen Safe talk about endocrine disruptors such...
by Elizabeth Kissling | Dec 29, 2009 | Activism, DIY, Humor, zine
If you’re anywhere near NYC, don’t miss this opportunity to see our friend Chella Quint perform tonight at Bluestockings. She will be reading from Issue #5 of Adventures in Menstruating, her awesome period-powered zine, at 7pm on Tuesday, December 29th, at...
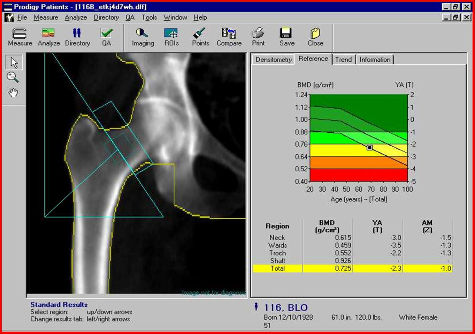
by Elizabeth Kissling | Dec 27, 2009 | Birth Control, New Research, Pharmaceutical
New research from the University of Texas Medical Branch at Galveston finds that nearly half of women using depot medroxyprogesterone acetate (DMPA), commonly known as the birth control shot, will experience high bone mineral density (BMD) loss in the hip or lower...
by Elizabeth Kissling | Dec 27, 2009 | Books, Health Care
One of the reviewers for the forthcoming edition of Our Bodies, Ourselves, who blogs anonymously at Mom’s Tinfoil Hat, is seeking contributions for the chapter she is reviewing. I am busy reviewing and contributing to the second chapter of Our Bodies, Ourselves that...
by Chris Hitchcock | Dec 24, 2009 | Birth Control, Dysmenorrhea, Health Care, Menorrhagia, Menstruation, Pharmaceutical, PMS
A recent press release from the American College of Obstetricians and Gynecologists announces that Hormonal Contraceptives Offer Benefits Beyond Pregnancy Prevention. This is in the same vein as similar articles published over the years about “non-contraceptive...




